All three binary pairs of components, nitromethane + 1-hexanol, nitromethane + water and 1-hexanol + water, are partially miscible. The data for first two systems were compiled and critically evaluated (Sazonov et al.4). The values of UCST (upper critical solution temperature) of the binary systems are reported in Zhuravleva et al.1 and Sazonov et al.2, and presented in Table 2. The 1-hexanol + water system was critically evaluated in Barton.5
The solubility temperatures of the liquid phases were determined by the synthetic method in ternary mixtures with constant mass relationship of (1) and (2), and also in ternary mixtures with a constant mass content of (3) (Zhuravleva et al.1). The solubility curves at 293.2 K, 294.2 K, 303.2 K and 313.2 K were determined by the titration method (Sazonov et al.2). Experimental errors were not reported, but are estimated by the evaluator to be ±0.1 K (temperature) and ±0.001 mass fractions (solubility). The system nitromethane + 1-hexanol + water includes a three-phase region. Depending on temperature the system nitromethane + 1-hexanol + water forms a miscibility gaps of Type I to III, Treybal.6 In the temperature range 273 K to 308.6 K, where three binary systems are partially miscible, the solubility curves correspond to Type III (Treybal6). A three-phase region occurs below 294.4 K (Zhuravleva et al.1). Maximal temperature of the three-phase liquid equilibrium was reported to be 294.2 K (Zhuravleva et al.1) and 294.5 K (Sazonov et al.2). Thus, the recommended value is Tc = (294.4 ±0.2) K. Two separate regions of immiscibility exist above 294.4 K, one of these can be described by band type curves. In the temperature range 308.6 K to 377.7 K (Zhuravleva et al.,1 Sazonov et al.2 and Sazonov et al.3), where two binary systems (nitromethane + water and 1-hexanol + water) are partially miscible, the solubility curves are Type II (Treybal6). Above 377.7 K, this ternary system has only one partially miscible region, Zhuravleva et al.1 Solubility data at 313 K and 333 K (Zhuravleva et al.1 and Sazonov et al.2) both separately and in common (only organic-rich branch) are described by an algebraic equation in the form:
This smoothing equation gives a smaller standard deviation than any other tested equation. The equation parameters (a, b, c, d and e) were calculated by the least squares method and presented in Table 3.
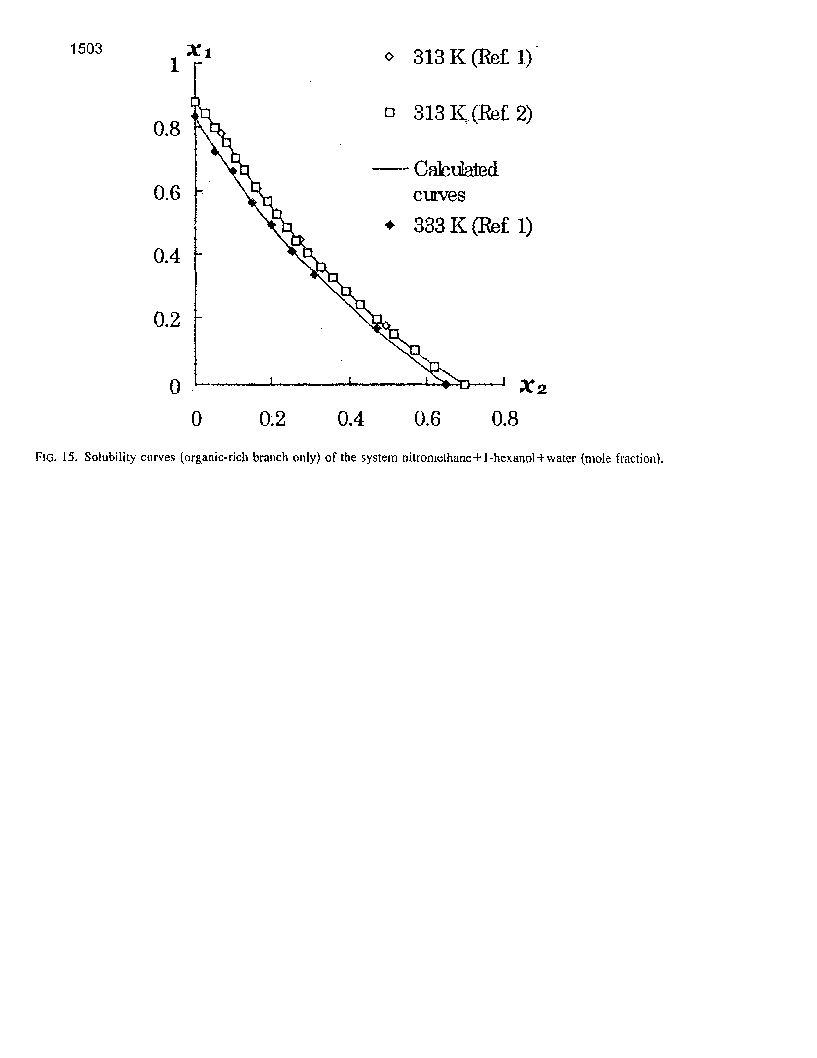
The solubilities, calculated by the proposed equations, are presented in Fig. 15 and in Table 4. Experimental solubility data, Zhuravleva et al.1 and Sazonov et al.2, are presented in Fig. 15 for comparison.
LLE data for the ternary system nitromethane + 1-hexanol + water were reported at 283 K, 291 K, 293 K and 298 K (Zhuravleva et al.1) in graphical form and at 294.2, 296.2 and 313.2 (Sazonov et al.3). Composition of each phase (Zhuravleva et al.1) was calculated from the index of refraction-composition curve of saturated mixtures prepared at the time of saturation curve measurements and by gas chromatography (Sazonov et al.3). The compositions of three coexisting liquid phases at 283 K, 291 K and 293 K (Zhuravleva et al.1) are also reported in graphical form. These data and data from Sazonov et al.3 at 294.2 K are presented in Table 5. Experimental errors were not reported. All experimental data are considered tentative.
Table 4. Calculated solubility data (organic-rich branch only) of nitromethane, 1-hexanol and water
| C T (K) | D w1 | D w2 | D w3 | D x1 | D x2 | D x3 |
|---|---|---|---|---|---|---|
| 313.2 | 0.0 | 9.29 x 10-1 | 7.1 x 10-2 | 0.0 | 6.98 x 10-1 | 3.02 x 10-1 |
| 313.2 | 2.9 x 10-2 | 8.95 x 10-1 | 7.6 x 10-2 | 3.5 x 10-2 | 6.50 x 10-1 | 3.15 x 10-1 |
| 313.2 | 6.5 x 10-2 | 8.54 x 10-1 | 8.1 x 10-2 | 7.6 x 10-2 | 6.00 x 10-1 | 3.24 x 10-1 |
| 313.2 | 1.05 x 10-1 | 8.09 x 10-1 | 8.6 x 10-2 | 1.20 x 10-1 | 5.50 x 10-1 | 3.30 x 10-1 |
| 313.2 | 1.52 x 10-1 | 7.59 x 10-1 | 8.9 x 10-2 | 1.68 x 10-1 | 5.00 x 10-1 | 3.32 x 10-1 |
| 313.2 | 2.05 x 10-1 | 7.04 x 10-1 | 9.1 x 10-2 | 2.20 x 10-1 | 4.50 x 10-1 | 3.30 x 10-1 |
| 313.2 | 2.65 x 10-1 | 6.43 x 10-1 | 9.2 x 10-2 | 2.76 x 10-1 | 4.00 x 10-1 | 3.24 x 10-1 |
| 313.2 | 3.31 x 10-1 | 5.77 x 10-1 | 9.2 x 10-2 | 3.36 x 10-1 | 3.50 x 10-1 | 3.14 x 10-1 |
| 313.2 | 4.04 x 10-1 | 5.07 x 10-1 | 8.9 x 10-2 | 4.01 x 10-1 | 3.00 x 10-1 | 2.99 x 10-1 |
| 313.2 | 4.85 x 10-1 | 4.31 x 10-1 | 8.4 x 10-2 | 4.71 x 10-1 | 2.50 x 10-1 | 2.79 x 10-1 |
| 313.2 | 5.71 x 10-1 | 3.50 x 10-1 | 7.9 x 10-2 | 5.46 x 10-1 | 2.00 x 10-1 | 2.54 x 10-1 |
| 313.2 | 6.64 x 10-1 | 2.66 x 10-1 | 7.0 x 10-2 | 6.26 x 10-1 | 1.50 x 10-1 | 2.24 x 10-1 |
| 313.2 | 7.62 x 10-1 | 1.79 x 10-1 | 5.9 x 10-2 | 7.12 x 10-1 | 1.00 x 10-1 | 1.88 x 10-1 |
| 313.2 | 8.63 x 10-1 | 9.0 x 10-2 | 4.7 x 10-2 | 8.03 x 10-1 | 5.0 x 10-2 | 1.47 x 10-1 |
| 313.2 | 9.65 x 10-1 | 0.0 | 3.5 x 10-2 | 8.90 x 10-1 | 0.0 | 1.10 x 10-1 |
| 333.2 | 0.0 | 9.14 x 10-1 | 8.6 x 10-2 | 0.0 | 6.52 x 10-1 | 3.48 x 10-1 |
| 333.2 | 3 x 10-3 | 9.11 x 10-1 | 8.6 x 10-2 | 4 x 10-3 | 6.50 x 10-1 | 3.46 x 10-1 |
| 333.2 | 3.9 x 10-2 | 8.70 x 10-1 | 9.1 x 10-2 | 4.5 x 10-2 | 6.00 x 10-1 | 3.55 x 10-1 |
| 333.2 | 8.0 x 10-2 | 8.25 x 10-1 | 9.5 x 10-2 | 8.9 x 10-2 | 5.50 x 10-1 | 3.61 x 10-1 |
| 333.2 | 1.25 x 10-1 | 7.75 x 10-1 | 1.00 x 10-1 | 1.35 x 10-1 | 5.00 x 10-1 | 3.65 x 10-1 |
| 333.2 | 1.77 x 10-1 | 7.20 x 10-1 | 1.03 x 10-1 | 1.85 x 10-1 | 4.50 x 10-1 | 3.65 x 10-1 |
| 333.2 | 2.35 x 10-1 | 6.60 x 10-1 | 1.05 x 10-1 | 2.38 x 10-1 | 4.00 x 10-1 | 3.64 x 10-1 |
| 333.2 | 2.98 x 10-1 | 5.95 x 10-1 | 1.07 x 10-1 | 2.94 x 10-1 | 3.50 x 10-1 | 3.56 x 10-1 |
| 333.2 | 3.70 x 10-1 | 5.24 x 10-1 | 1.06 x 10-1 | 3.55 x 10-1 | 3.00 x 10-1 | 3.45 x 10-1 |
| 333.2 | 4.48 x 10-1 | 4.47 x 10-1 | 1.05 x 10-1 | 4.19 x 10-1 | 2.50 x 10-1 | 3.31 x 10-1 |
| 333.2 | 5.33 x 10-1 | 3.66 x 10-1 | 1.01 x 10-1 | 4.88 x 10-1 | 2.00 x 10-1 | 3.12 x 10-1 |
| 333.2 | 6.26 x 10-1 | 2.79 x 10-1 | 9.5 x 10-2 | 5.63 x 10-1 | 1.50 x 10-1 | 2.87 x 10-1 |
| 333.2 | 7.26 x 10-1 | 1.89 x 10-1 | 8.5 x 10-2 | 6.43 x 10-1 | 1.00 x 10-1 | 2.57 x 10-1 |
| 333.2 | 8.31 x 10-1 | 9.5 x 10-2 | 7.4 x 10-2 | 7.31 x 10-1 | 5.0 x 10-2 | 2.19 x 10-1 |
| 333.2 | 9.44 x 10-1 | 0.0 | 5.6 x 10-2 | 8.30 x 10-1 | 0.0 | 1.70 x 10-1 |
Table 5. Compositions of three coexisting liquid phases system nitromethane + 1-hexanol + water
| C T (K) | D x1 | D x2 |
|---|---|---|
| 283.2 | 8.21 x 10-1 | 6.2 x 10-2 |
| 291.2 | 6.84 x 10-1 | 1.31 x 10-1 |
| 293.2 | 6.04 x 10-1 | 2.03 x 10-1 |
| 294.2 | 6.20 x 10-1 | 1.83 x 10-1 |
| 283.2 | 2.30 x 10-1 | 5.11 x 10-1 |
| 291.2 | 3.70 x 10-1 | 3.49 x 10-1 |
| 293.2 | 4.18 x 10-1 | 3.05 x 10-1 |
| 294.2 | 4.46 x 10-1 | 2.96 x 10-1 |
| 283.2 | 3.2 x 10-2 | 2 x 10-3 |
| 291.2 | 3.3 x 10-2 | 2 x 10-3 |
| 293.2 | 3.3 x 10-2 | 2 x 10-3 |
| 294.2 | 3.4 x 10-2 | 1 x 10-3 |
Figure
References:
1
Zhuravleva, I. K.; Zhuravlev, E. F.; Rudakova, N. A.; Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol. 18, 333 (1975).
2
Sazonov, V. P.; Markuzin, N. P.; Filippov, V. V.; Zh. Prikl. Khim. 49, 784 (1976).
3
Sazonov, V. P.; Markuzin, N. P.; Filippov, V. V.; Zh. Prikl. Khim. 50, 1524 (1977).
4
Barton, A. F. M.; 'Alcohols with Water' IUPAC Solubility Data Series, 1984, Vol. 15. Pergamon Press.
5
Treybal, R. E.; Liquid Extraction, McGraw-Hill, New York, 1963.
6
Sazonov, V. P.; Marsh, K. N.; Hefter, G. T.; Nitromethane with Water or Organic Solvents: Binary Systems, IUPAC-NIST Solubility Data Series. Vol. 71, J. Phys. Chem. Ref. Data 29 (2000).
