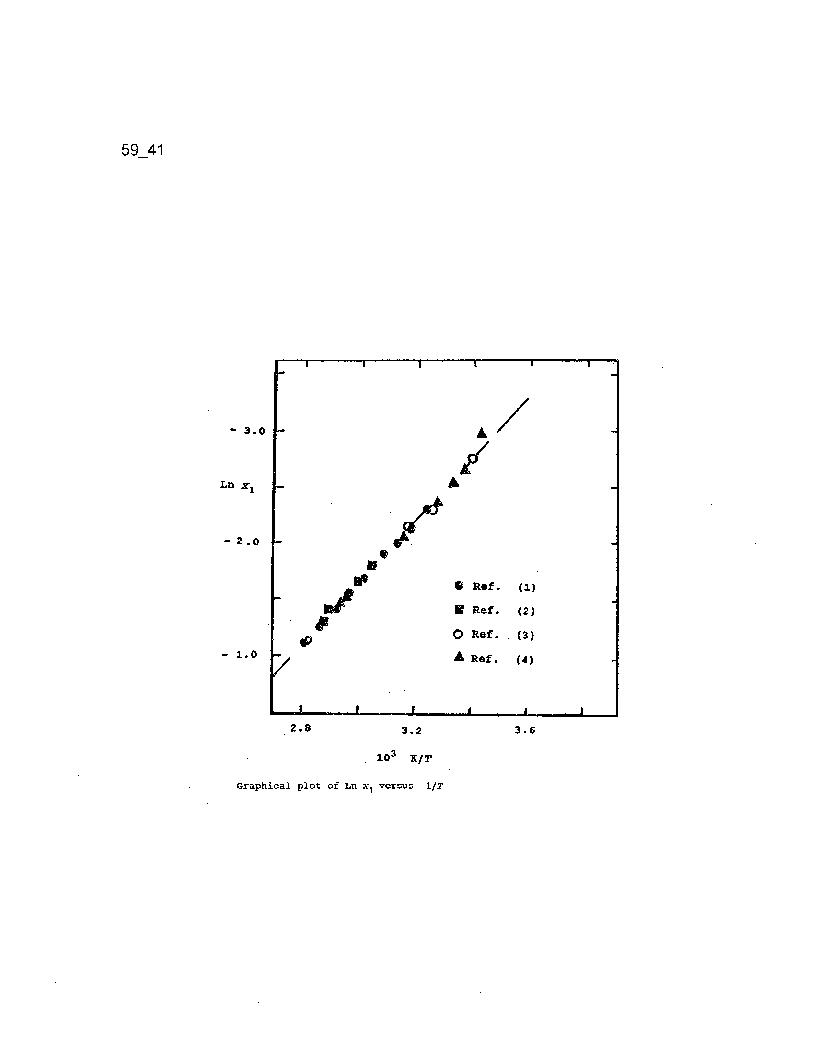
Regressional analysis of the experimental data as Ln x1 versus 1/T yielded the following mathematical relationship: Ln x1 = - 2668.6 (1/T) + 6.8891 (r=0.9995) for variation of fluorene solubility with absolute temperature (see graph).
Figure
References:
1
Choi, P. B.; Williams, C. P.; Buehring, K. G.; McLaughlin, E.; J. Chem. Eng. Data 1985, 30, 403-409.
2
McLaughlin, E.; Zainal, H. A.; J. Chem. Soc. 1959, 863-867.
3
Mortimer, F. S.; J. Am. Chem. Soc. 1923, 45, 633-641.
4
Domanska, U.; Groves Jr., F. R.; McLaughlin, E.; J. Chem. Eng. Data 1993, 38, 88-94.
