Components:
(1) Carbon dioxide,
CO2; [124-38-9]
NIST WebBook
(2) Poly(vinyl butyral) (PVBu),
None; [915977-69-4]
NIST WebBook
Original Measurements
Kamiya, Y.; Hirose, T.; Mizoguchi, K.; Terada, K.; J. Polym. Sci. B: Polym. Phys. 26, 1409 (1988).
Variables:
Ambient temperature: 298 K
Pressure: 0 - 5.0 MPa
Prepared by:
Method/Apparatus/Procedure:
Sorption isotherms were measured gravimetrically with a Cahn model 2000 electrobalance. A correction for the buoyancy was introduced.
Source and Purity of Materials:
- CO2: purity 99.99%.
PVBu Mitsubishi Monsanto Chemical Co., density at 308 K, 1.087 g/cm3, Mw between 34 000 and 38 000, butyral content 88%.
Estimated Errors:
Ambient temperature: No information given.
References
1 Hirose, T.; Mizoguchi, K.; Kamiya, Y.; J. Appl. Polym. Sci. 30, 401 (1985).
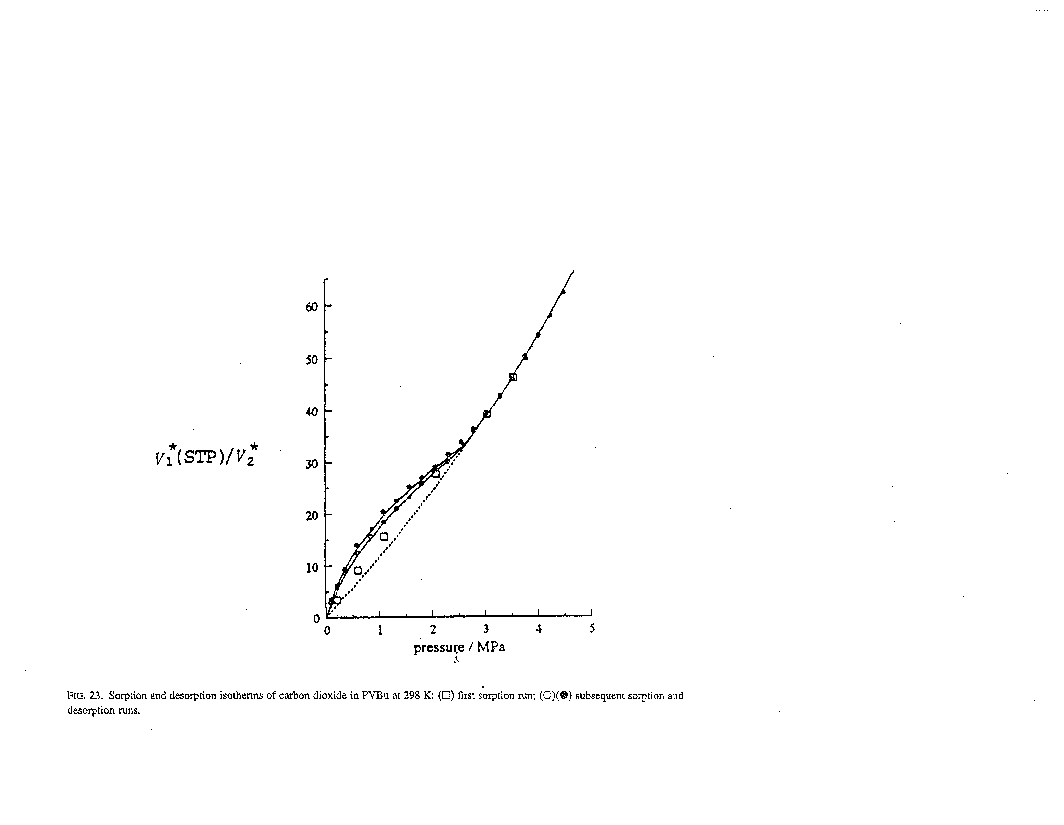
Experimental Values:
Note: The isotherm was treated by the equation:C=CD+CH=[kD exp(sC*)]p+C1HObp[(1-C*/Cg)/(1+bp)],
where the parameters kD, C1HO, and b have the usual meaning. Cg is the solute concentration corresponding to the glass transition and
C*=CD+fCH=[kDp+fC'HObp/(1+bp)]/[1+fC1HObp/Cg(1+bp)],
where f is an adjustable parameter, characterizing the plastisizing ability of a solute. Parameters for ordinary dissolution (Flory-Huggins's) and for Langmuir isotherms at 298K.
| β (cm3(g) cm-3(s) atm-1) | H (cm3(g) cm-3(s) atm-1) | C1 (cm3/cm3) | C'H (cm3/cm3) | b (atm-1) |
|---|---|---|---|---|
| 3.5 x 10-3 | 1.13 | 34.0 | 20.7,28.6 | 9.6 x 10-2 |
(a) Measurements are adsorption and desorption.
None



